Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect
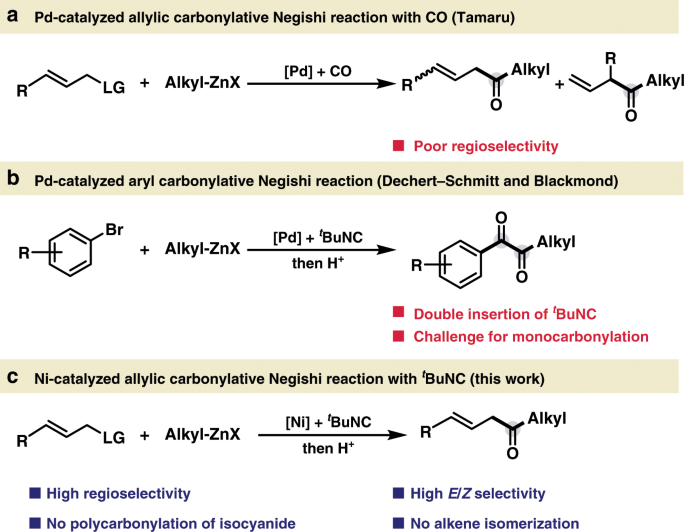
Nickel-catalyzed allylic carbonylative coupling of alkyl zinc reagents with tert -butyl isocyanide | Nature Communications
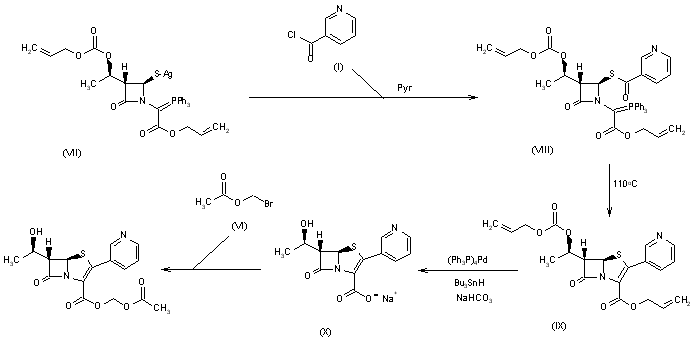
Research Letter A Novel, One-Step Palladium and Phenylsilane Activated Amidation from Allyl Ester on Solid Support
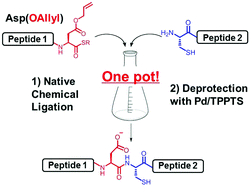
Efficient peptide ligation between allyl-protected Asp and Cys followed by palladium-mediated deprotection - Chemical Communications (RSC Publishing)
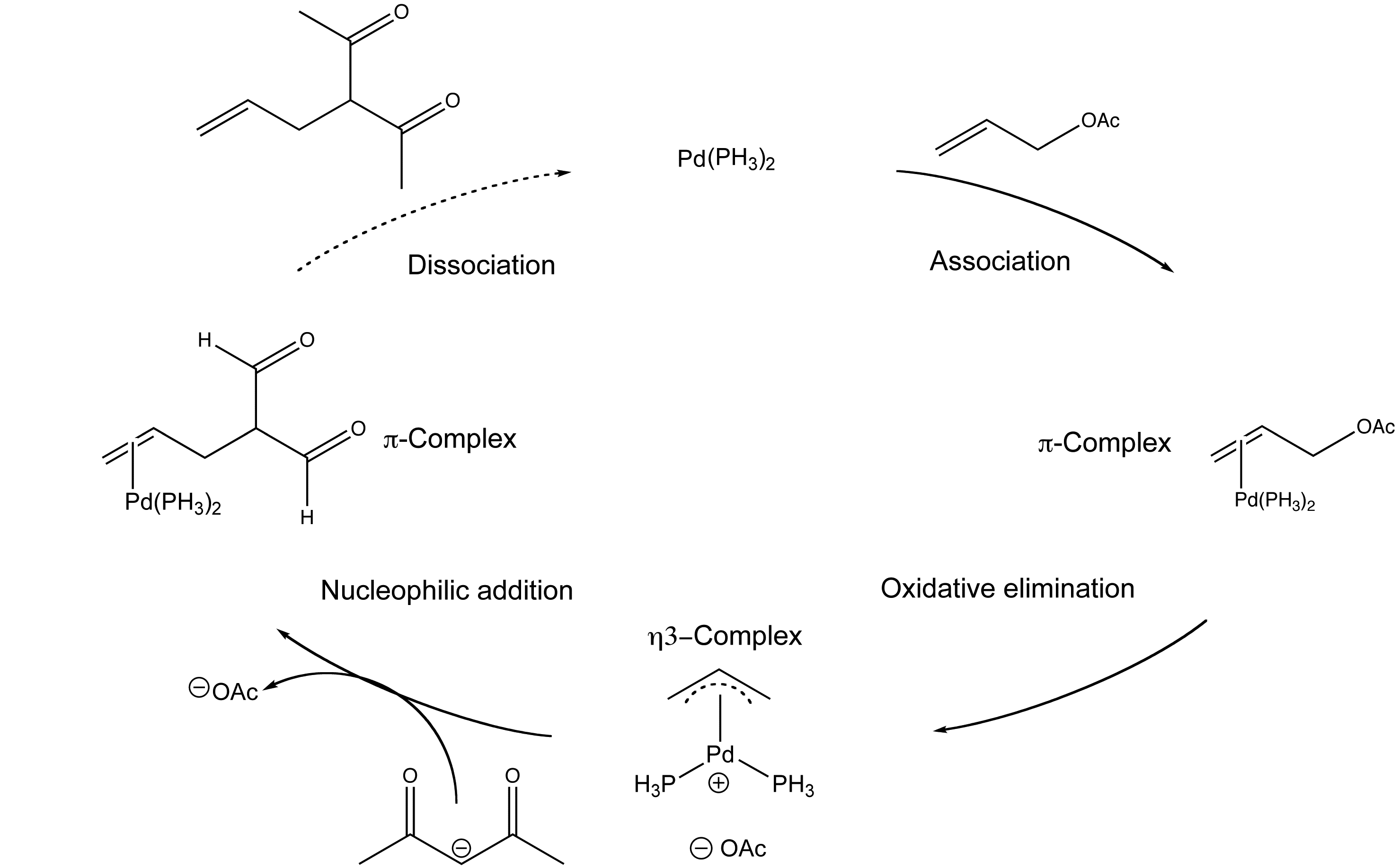
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds
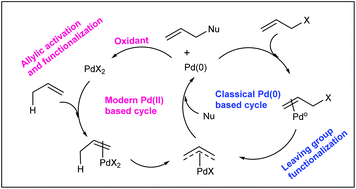
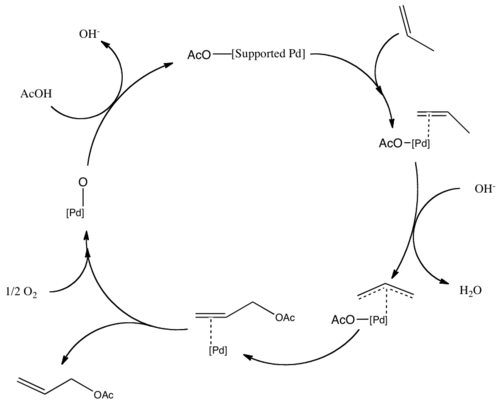
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)
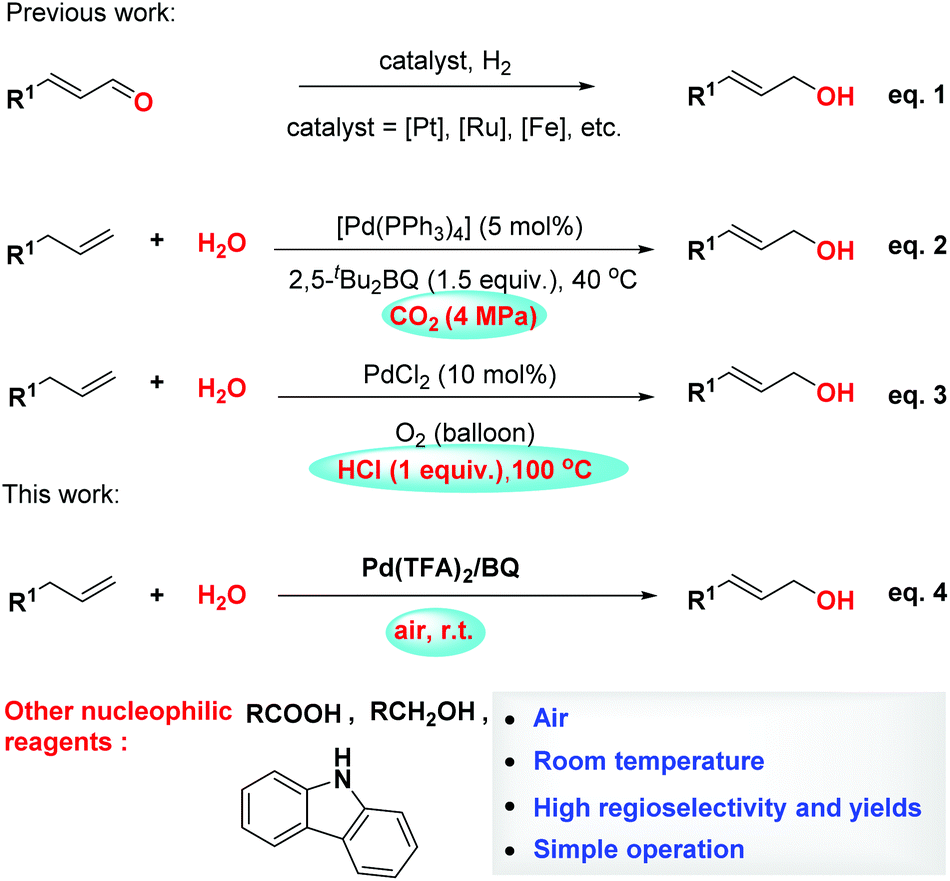
Palladium-catalyzed allylic C–H oxidation under simple operation and mild conditions - Organic & Biomolecular Chemistry (RSC Publishing)
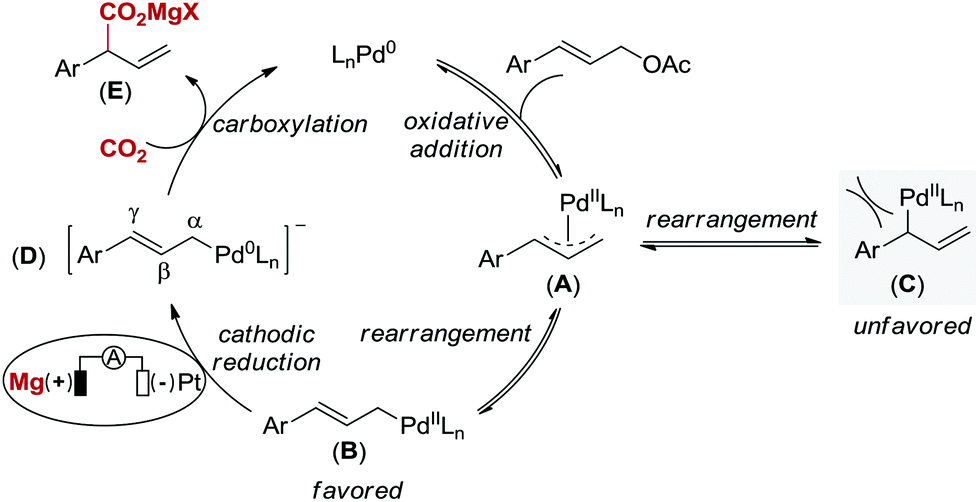
Palladium-catalyzed reductive electrocarboxylation of allyl esters with carbon dioxide - Organic Chemistry Frontiers (RSC Publishing)

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane-ZnCl2/Pd(PPh3)4
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions
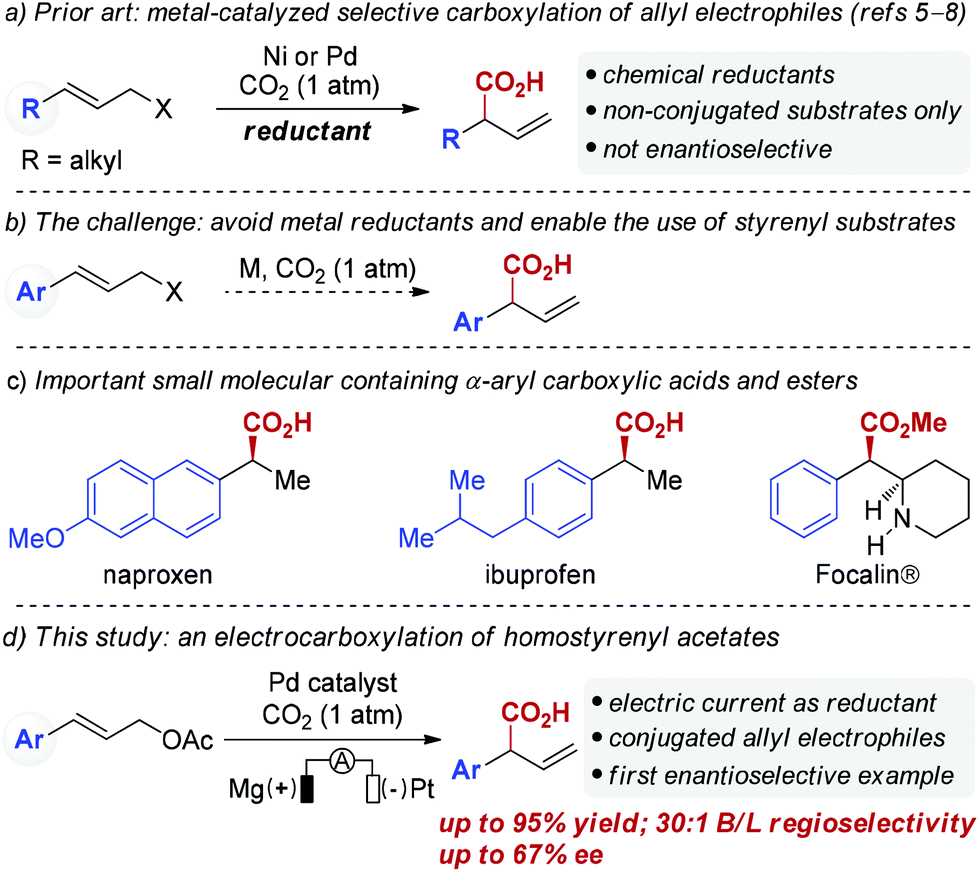
Palladium-catalyzed reductive electrocarboxylation of allyl esters with carbon dioxide - Organic Chemistry Frontiers (RSC Publishing)

Safe Removal of the Allyl Protecting Groups of Allyl Esters using a Recyclable, Low‐Leaching and Ligand‐Free Palladium Nanoparticle Catalyst - Takagi - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium(0)‐Catalyzed Rearrangement of Allylic Esters - Jessen - 2020 - ChemistrySelect - Wiley Online Library