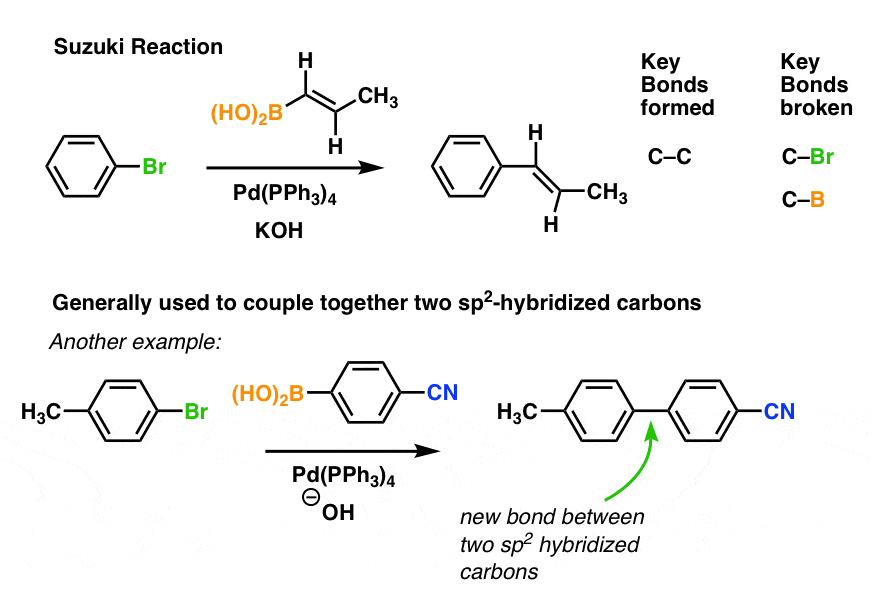
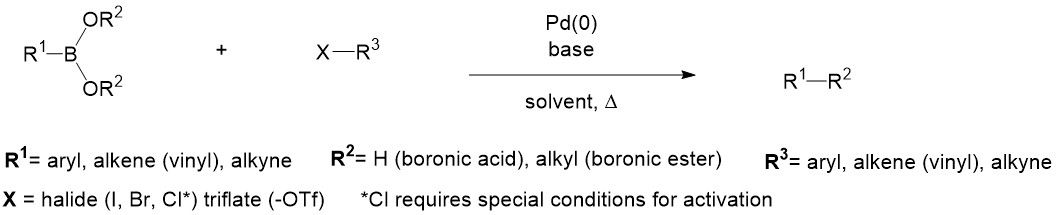
Suzuki-Miyaura cross-coupling of phenylboronic acid with aryl halides catalyzed by palladium and nickel species supported on alumina-based oxides - ScienceDirect

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and

PdCl2 Immobilized in Polyacrylamide: a Low Cost and Eco-Friendly Catalyst for Suzuki-Miyaura Reactions

Table 6 | Magnetic Mesoporous Silica Nanocomposite Functionalized with Palladium Schiff Base Complex: Synthesis, Characterization, Catalytic Efficacy in the Suzuki–Miyaura Reaction and α-Amylase Immobilization | SpringerLink

Figure 1 from Palladium-Catalyzed Synthesis of (Hetero)Aryl Alkyl Sulfones from (Hetero)Aryl Boronic Acids, Unactivated Alkyl Halides, and Potassium Metabisulfite. | Semantic Scholar
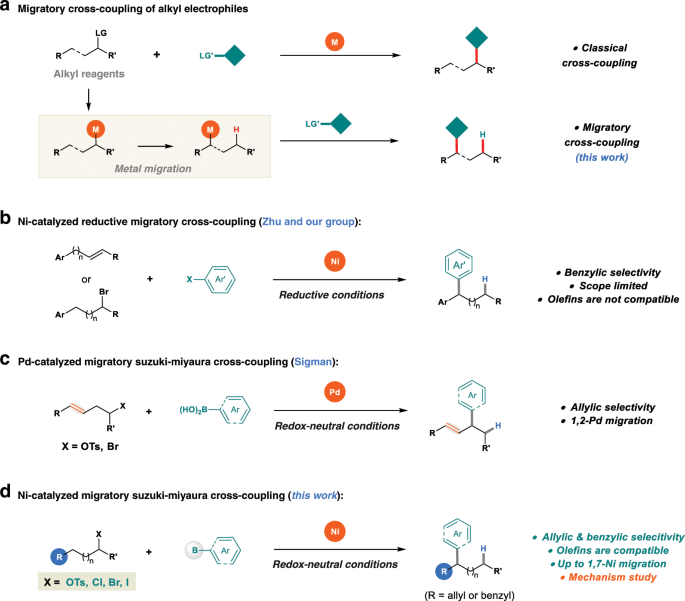
Reaction scope and mechanistic insights of nickel-catalyzed migratory Suzuki–Miyaura cross-coupling | Nature Communications

Palladium-Catalyzed, Direct Boronic Acid Synthesis from Aryl Chlorides: A Simplified Route to Diverse Boronate Ester Derivatives
PLOS ONE: Supported Palladium Nanoparticles Synthesized by Living Plants as a Catalyst for Suzuki-Miyaura Reactions

Palladium‐Catalyzed Direct Arylation of (Hetero)Arenes with Aryl Boronic Acids - Yang - 2008 - Angewandte Chemie International Edition - Wiley Online Library
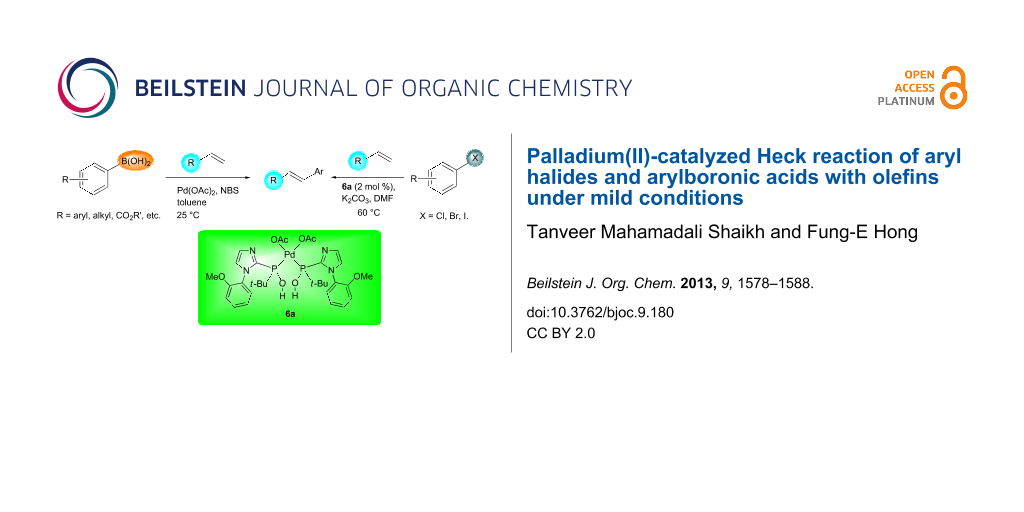
Palladium(II)-catalyzed Heck reaction of aryl halides and arylboronic acids with olefins under mild conditions

Table 1 from Fibrous nano-silica (KCC-1)-supported palladium catalyst: Suzuki coupling reactions under sustainable conditions. | Semantic Scholar

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect
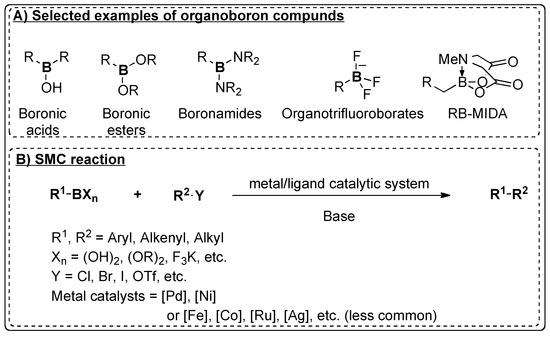
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML
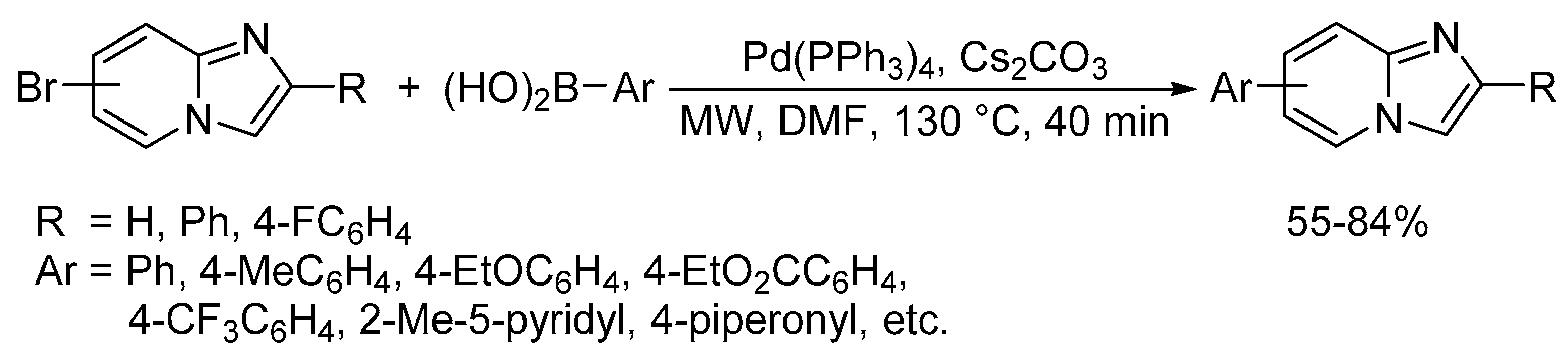
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Palladium-catalyzed cross-coupling reactions of aryl boronic acids with aryl halides in water - PDF Free Download

Palladium-catalyzed cross-coupling reactions of aryl boronic acids with aryl halides in water - PDF Free Download