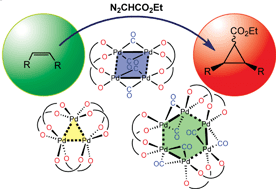
Palladium(II) carboxylates and palladium(I) carbonyl carboxylate complexes as catalysts for olefin cyclopropanation with ethyl diazoacetate - Dalton Transactions (RSC Publishing)

Direct Dehydrogenation for the Synthesis of α,β‐Unsaturated Carbonyl Compounds - Chen - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO) 6 as Carbonyl Source | SpringerLink

PDF) Palladium(I) and palladium(0) carbonyl bromide complexes | Vitaly Belsky and Oleg Temkin - Academia.edu
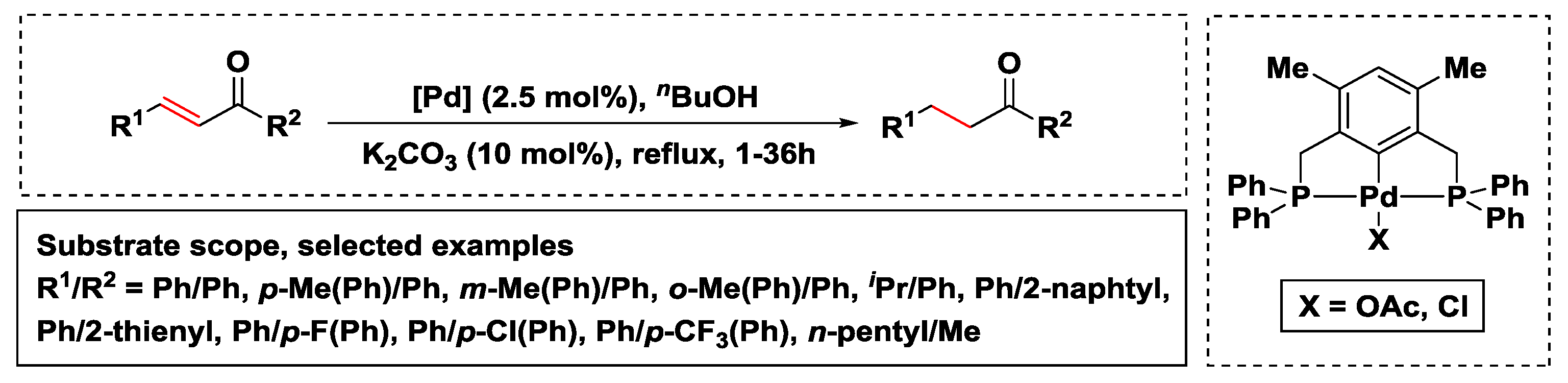
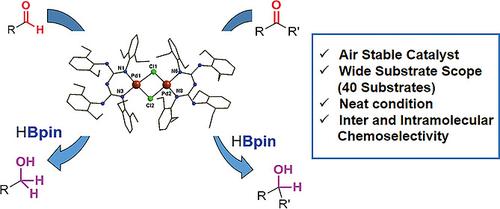
Catalysts | Free Full-Text | Hydrogen Transfer Reactions of Carbonyls, Alkynes, and Alkenes with Noble Metals in the Presence of Alcohols/Ethers and Amines as Hydrogen Donors | HTML
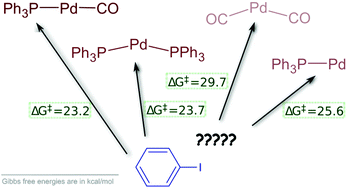
Viable pathways for the oxidative addition of iodobenzene to palladium (0)-triphenylphosphine-carbonyl complexes: a theoretical study - Dalton Transactions (RSC Publishing)

Synthesis, characterization and CO-releasing property of palladium(II) bipyridine flavonolate complexes | SpringerLink
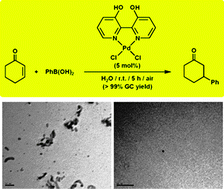
Conjugate addition of arylboronic acids to α,β-unsaturated carbonyl compounds in aqueous medium using Pd(ii) complexes with dihydroxy-2,2′-bipyridine ligands: homogeneous or heterogeneous nano-catalysis? - Catalysis Science & Technology (RSC Publishing)

Diastereoselective Carbonyl Allylation with Simple Olefins Enabled by Palladium Complex-Catalyzed C-H Oxidative Borylation
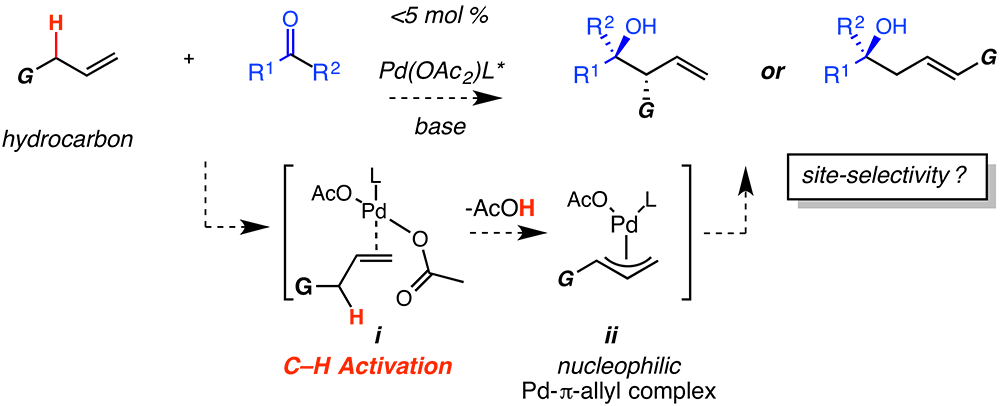
Report: Metal Catalyzed C-H Bond Activation and Allyl Addition: Development of New Transition Metal Catalysis for Practical and Uniquely Efficient Carbon-Carbon Bond Forming Reactions (58th Annual Report on Research Under Sponsorship of

Selective deoxygenation of carbonyl groups at room temperature and atmospheric hydrogen pressure over nitrogen-doped carbon supported Pd catalyst - ScienceDirect

Pyrazol-1-yl)carbonyl palladium complexes as catalysts for ethylene polymerization reaction - ScienceDirect
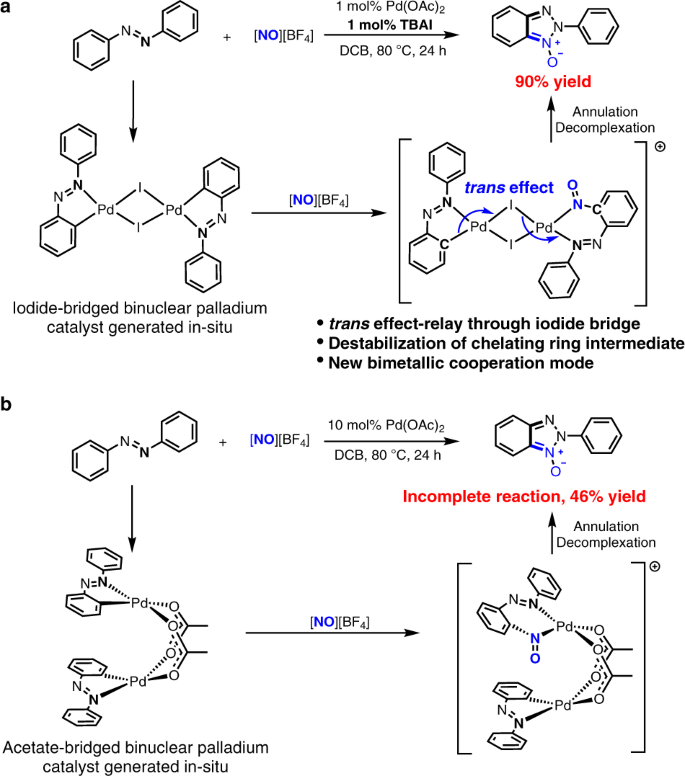
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
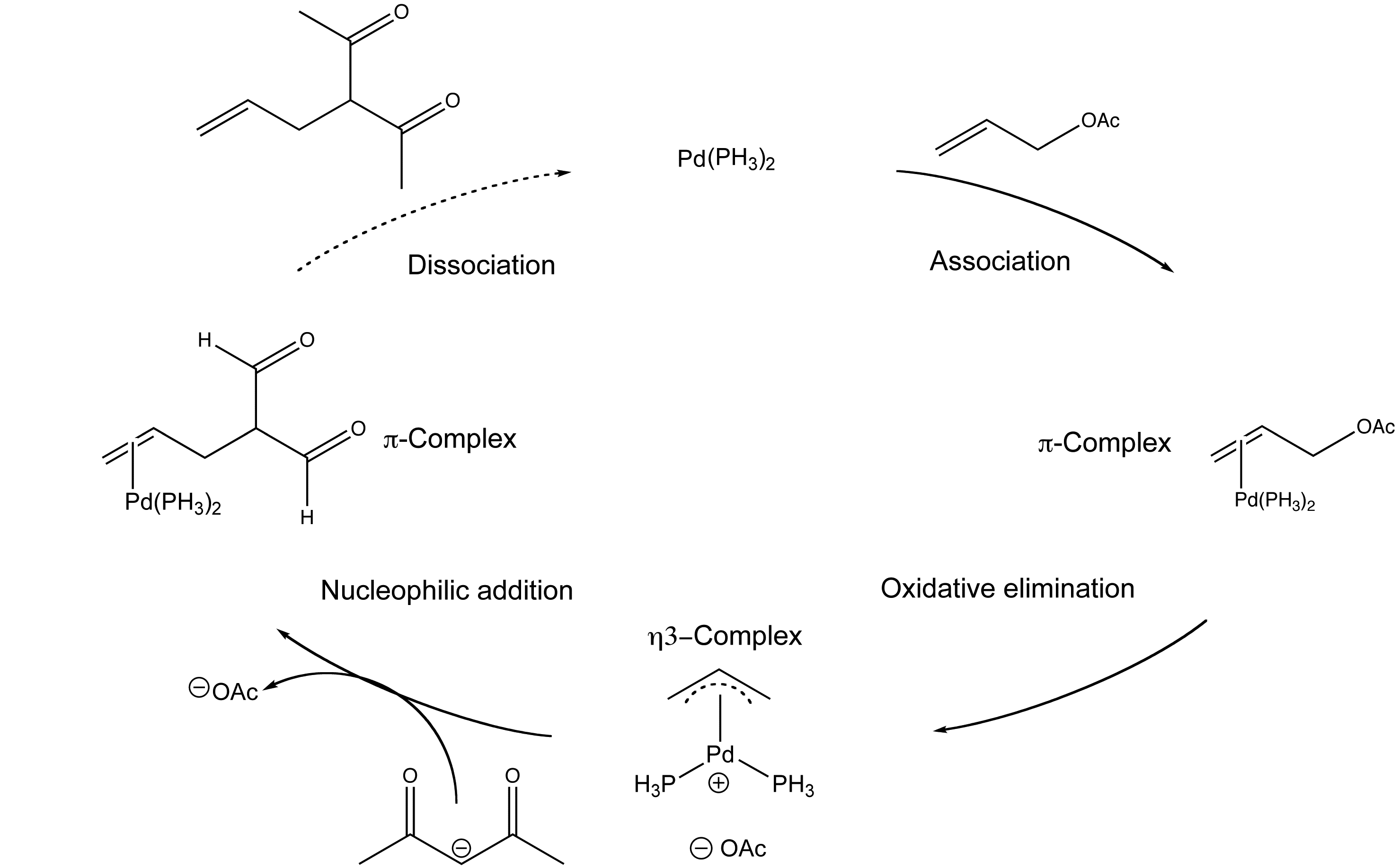
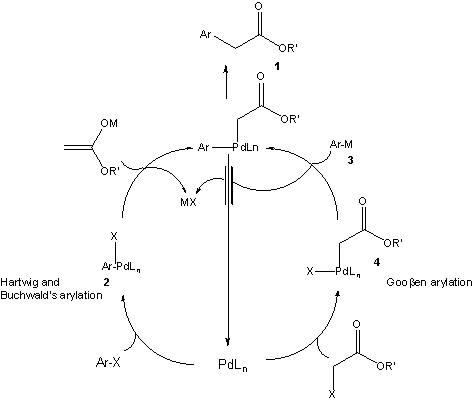
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

PDF) Hydrogenation of α,β-Unsaturated Carbonyl Compounds Using Recyclable Water-Soluble FeII/EDTA Complex Catalyst
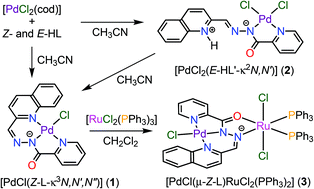
Palladium(ii) mononuclear and palladium(ii)/ruthenium(ii) heterodinuclear complexes containing 2-quinolyl-substituted (pyridine-2-carbonyl)hydrazone - Dalton Transactions (RSC Publishing)

Figure 3 from A palladium(II) complex containing both carbonyl and imine oxime ligands: crystal structure, experimental and theoretical UV-vis, IR and NMR studies. | Semantic Scholar

Catalysts | Free Full-Text | Hydrogen Transfer Reactions of Carbonyls, Alkynes, and Alkenes with Noble Metals in the Presence of Alcohols/Ethers and Amines as Hydrogen Donors | HTML



![IR carbonyl stretching frequencies of [Rh(CO) 2 Cl(L)] complexes... | Download Table IR carbonyl stretching frequencies of [Rh(CO) 2 Cl(L)] complexes... | Download Table](https://www.researchgate.net/publication/309115781/figure/tbl1/AS:667603408285696@1536180464134/IR-carbonyl-stretching-frequencies-of-RhCO-2-ClL-complexes-bearing-various.png)