
What can be done? Digital Media Policy Options for Europe (and beyond) | Reuters Institute for the Study of Journalism
B2 – Analytical report on EU law applicable to sharing of non-personal data Support Centre for data sharing

Improving the Safety of Medicines in the European Union: From Signals to Action - Potts - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library
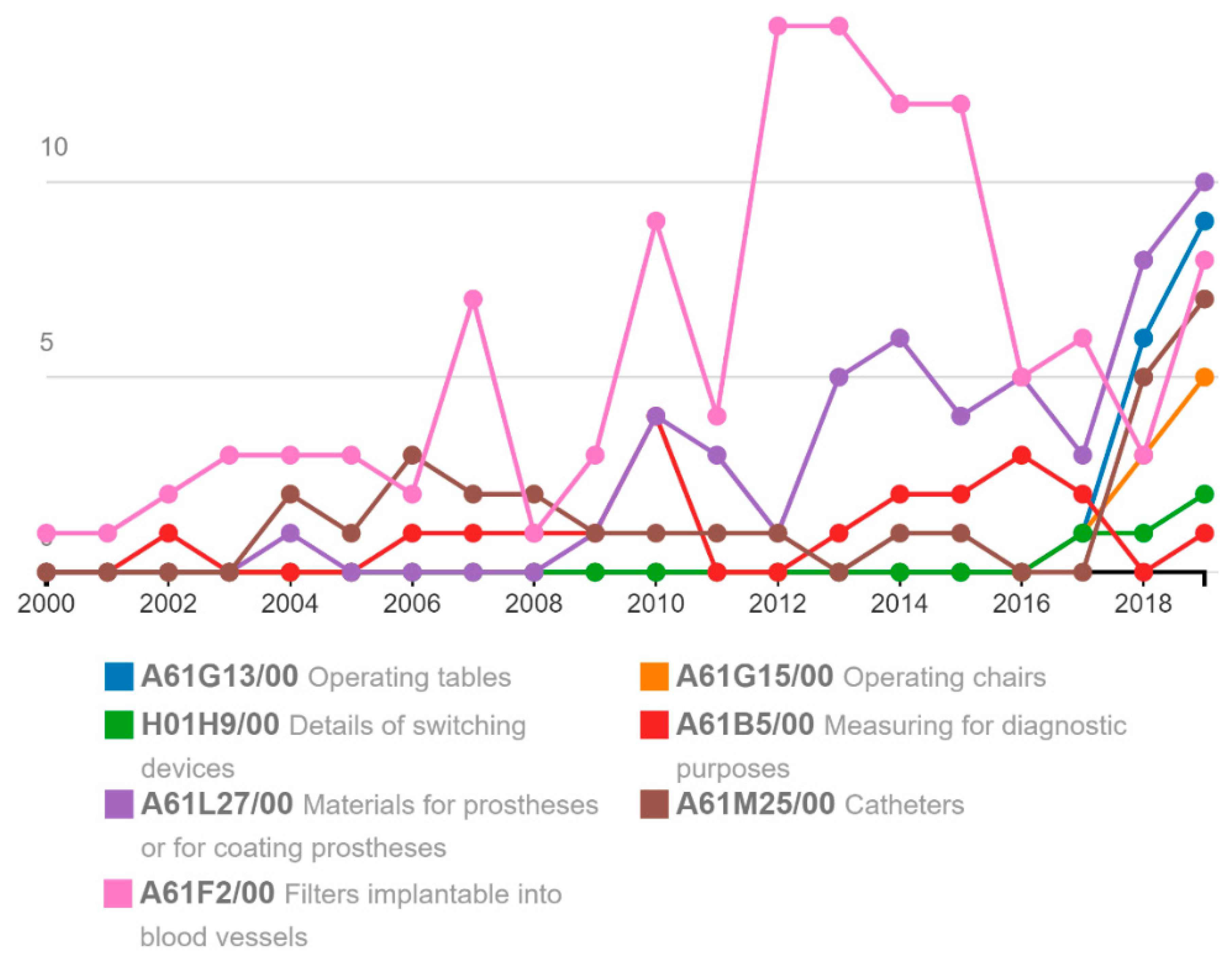
Administrative Sciences | Free Full-Text | New Regulations on Medical Devices in Europe: Are They an Opportunity for Growth? | HTML
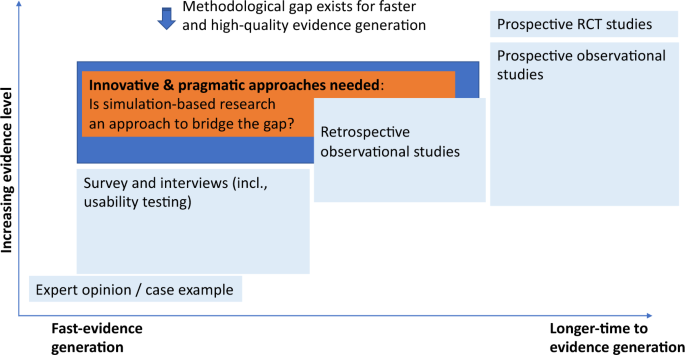
Challenges for the evaluation of digital health solutions—A call for innovative evidence generation approaches | npj Digital Medicine

Conducting Non-COVID-19 Clinical Trials during the Pandemic: Can Today's Learning Impact Framework Efficiency? in: European Journal of Health Law Volume 27 Issue 5 (2020)
B2 – Analytical report on EU law applicable to sharing of non-personal data Support Centre for data sharing











