PDF) Phase-Separable Polyisobutylene Palladium-PEPPSI Precatalysts: Synthesis and Application in Buchwald– Hartwig Amination
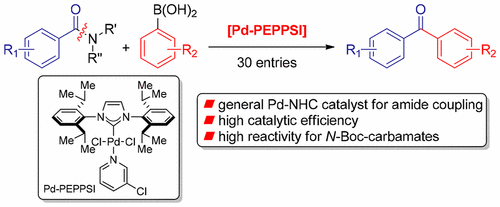
Pd-PEPPSI: Pd-NHC Precatalyst for Suzuki–Miyaura Cross-Coupling Reactions of Amides - J. Org. Chem. - X-MOL
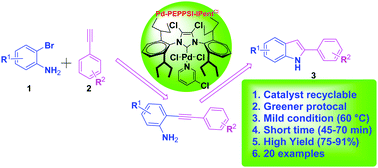
Pd-PEPPSI-IPentCl: a new highly efficient ligand-free and recyclable catalyst system for the synthesis of 2-substituted indoles via domino copper-free Sonogashira coupling/cyclization - New Journal of Chemistry (RSC Publishing)
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink
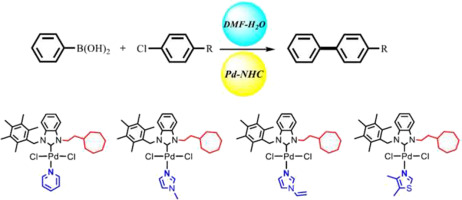
Cycloheptyl substituted N-heterocyclic carbene PEPPSI-type palladium complexes with different N-coordinated ligands: Involvement in Suzuki-Miyaura reaction - J. Organomet. Chem. - X-MOL

Synthesis of N-heterocyclic carbene-palladium-PEPPSI complexes and their catalytic activity in the direct C-H bond activation - ScienceDirect

PEPPSI‐Type Palladium–NHC Complexes: Synthesis, Characterization, and Catalytic Activity in the Direct C5‐Arylation of 2‐Substituted Thiophene Derivatives with Aryl Halides - Kaloğlu - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library
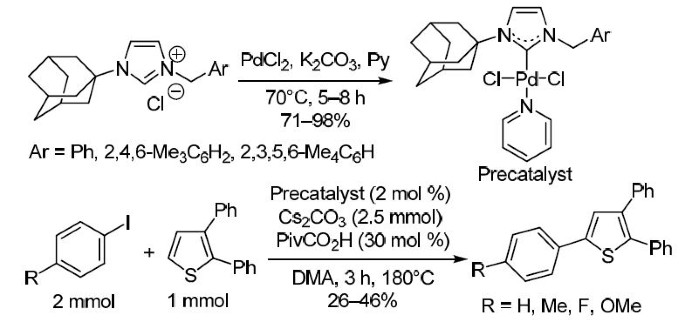
Adamantanyl-substituted PEPPSI-type palladium(II) N-heterocyclic carbene complexes: synthesis and catalytic application for CH activation of substituted thiophenes | SpringerLink

Synthesis, characterization and catalytic activity of PEPPSI-type palladium–NHC complexes - ScienceDirect

Investigation of activity, stability, and degradation mechanism of surface-supported Pd-PEPPSI complexes for Suzuki-Miyaura coupling - ScienceDirect
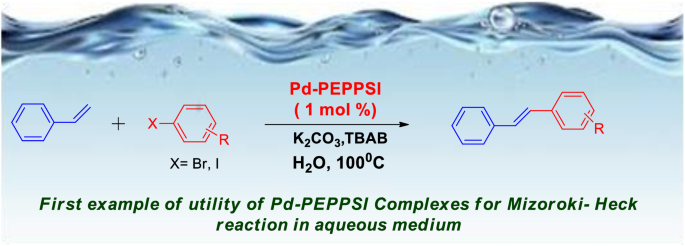
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink
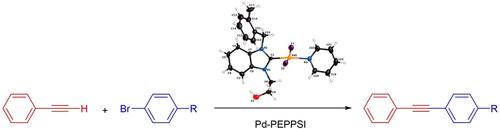
2‐hydroxyethyl‐substituted (NHC)PdI2(pyridine) (Pd‐PEPPSI) Complexes: Synthesis, Characterization and the Catalytic Activity in the Sonogashira Cross‐coupling Reaction,ChemistrySelect - X-MOL

Alternative Synthetic Methods for PEPPSI‐Type Palladium Complexes - Green - 2014 - European Journal of Inorganic Chemistry - Wiley Online Library
![Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML](https://www.mdpi.com/catalysts/catalysts-10-01081/article_deploy/html/images/catalysts-10-01081-g001.png)
Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML

Figure 1 from PEPPSI-type palladium complexes containing basic 1,2,3-triazolylidene ligands and their role in Suzuki-Miyaura catalysis. | Semantic Scholar

Synthesis of bridged palladium-PEPPSI complexes and catalytic studies in C–C cross-coupling reactions - ScienceDirect
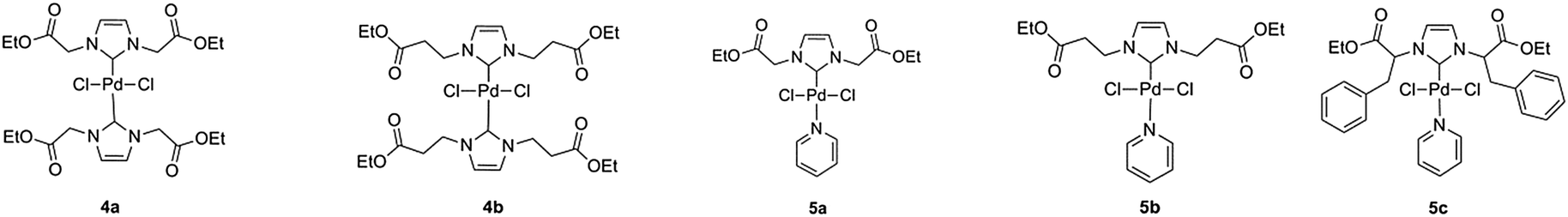
Amino acid-derived N-heterocyclic carbene palladium complexes for aqueous phase Suzuki–Miyaura couplings - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03337C



![Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram](https://www.researchgate.net/profile/Muhammad_Ghufran_Rafique/publication/317660984/figure/fig1/AS:528262394638337@1502958977918/Various-NHC-Pd-PEPPSI-left-and-long-alkyl-chain-labeled-NHC-Pdallyl-cinnamyl.png)